
The Use of Nuclear powered batteries has been for around more than a century. however, they promise in power that they sometimes lose in bulk.
It is not so with a new type of power source, which combines a novel structure with a nickel isotope to pack ten times more power than an electrochemical cell. The only question is, are we ready to end the atom?
A team of Russian researchers has introduced a new spin on technology that uses beta decay of a radioactive element to create a difference in the voltage.
To make a difference in the capacity of the voltage, your average electromechanical cell uses contrasts in response to various materials.
It gives a lot of power to the amount of battery, especially when using lithium-like material.
But as we all know well, the battery based on electrochemistry is not enough. They need to recharge or replace sooner or later.
On the other hand, the lifetime of an atomic battery is not based on its responsiveness, but it has a half-life of decay. Rather than measured in hours or days, their possible lifespan may be decades or centuries.
In 1913 the so-called Betawalic Batteries were dreamed. They were not like the small nuclear reactors. Instead of producing heat, they get their charge from beta particles emitted by an isotope-biting electron from another material.
Unfortunately, it does not really result in a torrential flood of electricity. To overcome this shortcoming, the movements of electrons can be fed into a capacitor like a capacitor. It works, but it also adds to the overall bulk.
Looking at this type of technique, you will be ideal for things you do not (or do not want to) to use – such as pacemakers or small satellites – the added mass is something you want to avoid.
What’s more, the word ‘radioactive’ is not as attractive as it was a century ago. In today’s world, nuclear-powered pacemaker marketing will demand more than an attractive jingle; But this new method can help to tip the scales.
The equipment is made with a specially made pair of nickel-63 isotopes between a pair of semiconductor diodes, called the Schottky barrier.
Betavoltaic cell
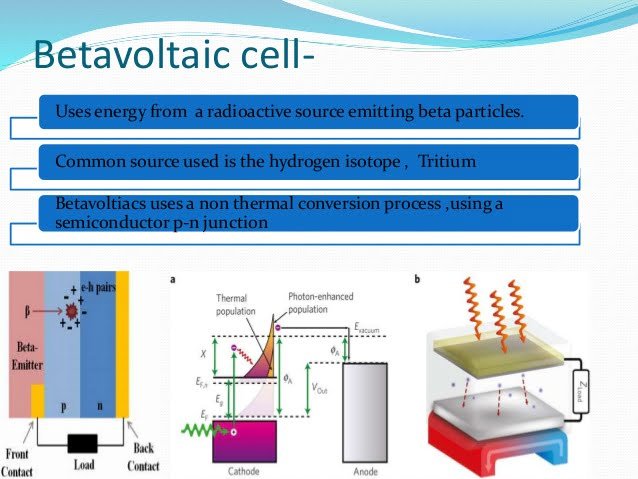
This barrier keeps the current in a way, a feature that is often used to convert alternate currents into direct.
Given that the optimum thickness of each layer was only 2 micrometers, researchers were able to maximize the voltage produced by each gram of isotope.
In Nickel-63, there is the only half-life of more than 100 years, which adds 3,300 millimeters per hour energy per gram in a customized manner like this: Ten times the specific energy of your particular electrochemical cell.
This is an important step from the last nickel-63 betavoltaic devices, and when it is not enough to power your smartphone, it brings it to be useful for various types of work.
“The higher the power density of the device,” says the Instrumental Institute for Superhard and Novell Carbon Materials, director of Vladimir Blanc, the more applications he has.
For example, the current generation of pacemakers is approximately 10 cubic centimeters in size, and about 10 microwaves power is provided by a subcutaneous electric chemical battery.
Wireless charging through the skin may be a possibility of one day, but for now, they need to be replaced by an invasive surgical operation.
Pacemakers will be completely simple and safe if they have a power source that exits the patient, and thus a betavoltaic cell can be ideal.
But after an initial love affair with all the radioactive things, a public appeal was flipped. The fear of half-century and the consequences of dealing with the threat of nuclear war has created confusion and distrust in the power of isotope.
Even communicating the risk of radioactivity using innocuous comparisons is very little to reduce the concerns on the potential impact of any of the materials described in the form of radioactive.
But for some, there will be technology to invest in small nuclear-powered equipment.



